After de-airing, the clamps were removed, and the ACP was converted to full flow bypass. Finally, the root was enlarged with a patch of homograft in the opened noncoronary aortic sinus, followed by homograft arch augmentation into the existing native tissue greater curvature scaffold. The proximal anastomosis was made between the fileted ascending aorta and the aortic root. The diameter of the distal opening connected to the native bridge tissue was checked. The posterior distal wall anastomosis was completed to form a native tissue bridge, which then formed the neo-greater arch curvature. The resulting bridge flap of native ascending aorta was rotated towards the right shoulder and anchored to the descending aorta. The ascending aorta was then fileted open with two parallel incisions all the way up to the proximal arch. All remnant ductal tissue was excised from the descending aorta. The aortic valve was probed to confirm size adequacy for the anticipated biventricular repair. Therefore, the aorta was transected at the sinotubular junction, and the root was opened down into the noncoronary sinus. The baby whose repair is shown in this video had a diminutive aortic root and 5 mm aortic valve. The right descending aorta was clamped, the head vessels were snared, and antegrade cerebral perfusion (ACP) was commenced at 50 cc/kg/minute (and adapted up to 70 cc/kg/min as needed using near-infrared spectroscopy monitoring). With a Gore-Tex graft to the innominate artery for arterial inflow and initial single venous right atrial cannulation, bypass was commenced, the distal ductus was cannulated then transected proximal to the cannula, and the patient was cooled to 25☌.Īfter cross-clamping and cardioplegia administration, the clamp was replaced with a hemoclip and the cardioplegia needle was removed to free up the field. In a neonate with type A interruption, right arch, right descending aorta, and aberrant left subclavian artery, a complete biventricular repair was planned. It provides a tension-free native tissue bridge with potential for growth, as well as providing a scaffold for patch augmentation in biventricular hearts or for Norwood stage I in univentricular palliation, which the authors performed in eight neonates/infants. The PDA and the VSD are both closed in the same procedure.In interrupted aortic arch with a wide interruption gap between proximal and distal aortic portions, the ascending aortic slide is a safe and reproducible technique. Repair is accomplished through a median sternotomy, and the arch may be reconstructed under deep hypothermic circulatory arrest, using a piece of homograft for augmentation if necessary. DiGeorge syndrome is present about 27% of the time and may be indicated by hypocalcemia. Echocardiography is used for diagnosis and can identify the site of the interruption as well as an anomalous right subclavian artery and any associated cardiac defects, including the VSD. Hyperventilation should be avoided so as to not increase pulmonary blood flow and further worsen systemic perfusion. 
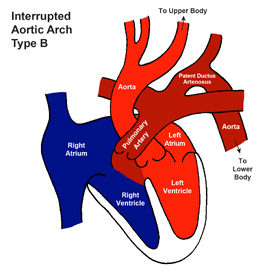
PGE1 should be infused to maintain ductal patency. There is eventually circulatory collapse and profound shock. The lower extremities may also become mottled or gray. As PVR decreases and pulmonary blood flow increases, patients develop signs of congestive heart failure, and these worsen as the ductus begins to close. Patients present within the first 2 weeks of life. Blood reaches the descending aorta via the ductus arteriosus (PGE-1 dependent). An aberrant right subclavian artery arising from the descending thoracic aorta and passing posterior to the esophagus is common, especially in type B. Most common type is B, where the defect lies between the left carotid and left subclavian artery. There are three types of IAA, and each describes where the interruption takes place. In interrupted aortic arch, there is an absence of luminal continuity between the ascending aorta and the descending aorta. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
